Navigate BioPharma Services AML-MRD Method Summary
Intended Use
The Acute Myeloid Leukemia Measurable Residual Disease by Multiparametric Flow Cytometry (AML MRD MFC) Test is a quasiquantitative, laboratory developed, in vitro test that utilizes consensus cell differentiation markers recommended by European Leukemia Network (ELN) to evaluate the presence of Leukemia Associated Immunophenotypes (LAIP) in the bone marrow (primarily) and peripheral blood (alternative when no bone marrow can be collected), of AML patients. AML-MRD MFC Test data collected from global clinical trials is interpreted centrally at Navigate BP CLIA Laboratory (a Novartis subsidiary), located in Carlsbad, CA. Navigate’s validation package fulfills Clinical and Laboratory Standards Institute (CLSI H62) guidelines commensurate with Secondary Clinical Trial Endpoint Data Submission and Patient Selection under CLIA (Tettero et al., 2023). AML-MRD MFC Test results in conjunction with standard clinical response criteria could be utilized to interpret the efficacy of investigational drugs for AML or High Risk-Myeloid Dysplastic Syndrome patients enrolled in clinical trials.
Disease Background & Method Rationale
AML is a cancer of the blood and bone marrow characterized by the rapid growth of abnormal white blood cells, known as myeloid blasts, which outgrow healthy blood cells resulting in bone marrow failure (Dohner et al., 2015). AML is often considered to be the most complex type of blood cancer due to its heterogenous morphology, immunophenotype, cytogenetics, molecular aberrations and poor survival outcomes following current approved treatment modalities (Hou & Tien, 2020). Indeed, the five-year survival rate is less than 30% for older patients (over 60 years) predominantly due to relapse highlighting the need for early detection of minimal residual disease (MRD) to guide therapy (Ossenkoppele and Schuurhuis, 2014; Short et al., 2018).
The most popular method for monitoring MRD in AML is the use of multiparametric flow cytometry (MFC), which can identify and quantify residual leukemic cells in the bone marrow or peripheral blood sample at a sensitivity of 0.1% or 1 in 1000 white blood cells (Buccisano et al., 2017). MRD status is prognostic for overall survival (OS) and relapse-free survival (RFS) (Schuurhuis et al., 2018; Short et al., 2020). MRD has gained importance not only in treatment decision-making but also in patient selection for clinical trials (Hu et al., 2020; Lowenberg et al., 2021). More importantly, MRD is expected to become a surrogate endpoint for survival based on industry wide meta-analyses, potentially expediting new drug approvals in the future. There is a growing emphasis on obtaining accurate MRD results and robust validation of MFC-based monitoring methods. Currently, MFC-MRD assays vary among different centers and depend on extensive scientific expertise and knowledge of inter-patient differences for accurate interpretation of data (Keeney et al., 2015). To harmonize interpretation and reporting, the European Leukemia Network (ELN) MRD working party on AML has recommended the utilization of a single consensus panel of markers for MRD monitoring (Heuser et al., 2021).
MFC Method Overview
Navigate’s AML-MRD MFC Assay is a 4-tube, 8-parameter assay incorporating cell differentiation markers for identification of the broadest group (>90%) of AML LAIPs (see Table 2). These markers are aligned with consensus recommendations from ELN AML MRD working party (Heuser et al., 2021; Schuurhuis et al., 2018), while specimen testing and interpretation principles are described in Cloos et al., 2018. Real world performance of this method has been evaluated in a multicenter clinical trial (Terwijn et al., 2013). A minimum of 500,000 white blood cells (WBCs) are acquired for each specimen on a Flow Cytometer System. LAIPs are identified utilizing a consistent DfN (different from normal) approach at baseline and tracked through the course of the treatment by trained and qualified scientists with extensive prior clinical experience in diagnosing myeloid neoplasms. Utilizing this approach, all the data from multi-center clinical studies are interpreted centrally at Navigate BP CLIA laboratory and test results are reported per ELN AML working party guidelines (Schuurhuis et al., 2018).
Table 2: AML-MRD MFC Assay Design
| LAIP Tube | Marker 1 | Marker 2 | Marker 3 | Marker 4 | Marker 5 | Marker 6 | Marker 7 | Marker 8 |
| LAIP Tube # 1 | CD34 | CD117 | HLA-DR | CD13 | CD45 | CD7 | CD56 | CD33 |
| LAIP Tube # 2 | CD34 | CD117 | HLA-DR | CD13 | CD45 | CD15 | CD22 | CD19 |
| LAIP Tube # 3 | CD34 | CD117 | HLA-DR | CD13 | CD45 | CD36 | CD14 | CD11b |
| LAIP Tube # 4 | CD34 | CD117 | HLA-DR | CD13 | CD45 | CD2 | CD133 | CD33 |
| Viability & Hemodilution | 7-AAD | - | - | CD13 | CD45 | CD15 | CD16 | - |
Patient Specimen Handling
Bone marrow aspirates and peripheral blood are collected in sodium-heparin tubes and shipped to the Navigate BP clinical testing laboratory at ambient (18-22 OC) temperature. All specimens received are visually inspected and a description of sample integrity, including clotting and/or hemolysis are documented. Hemodilution is monitored using CD13 versus CD16 stain in the granulocyte gate outlined in Table 2 above. Ambient specimen stability is 3 days; specimens received outside the stability window are evaluated by a board-certified pathologist and results are reported with a “disclaimer”. Specimen handling workflow is illustrated in Figure 1 and representative patient results are presented in Table 3.
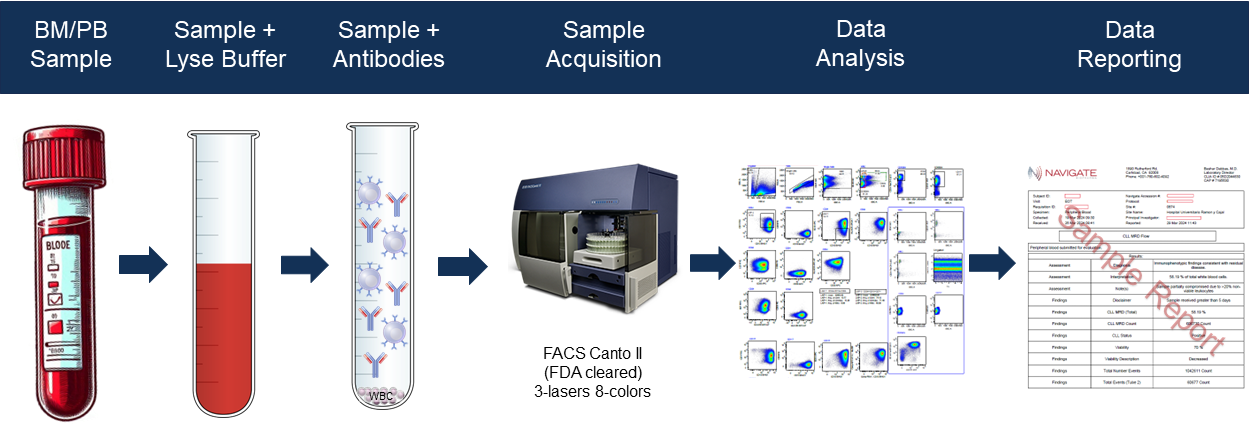
Figure 1. AML-MRD MFC Assay Workflow
Interpretation Principles
The general principles of our gating strategy are illustrated in Figure 2 below and described in Cloos 2018. The first three gates in the analysis scheme (Cells, Single Cells and WBC) help in eliminating cell debris, dead cells, erythrocytes and gate on white blood cell population. Lymphocyte, Monocyte and Granulocyte gates are placed by backgating cell lineage specific markers including CD7, CD19, CD13, HLADR and CD33. CD45dim gate is a flexible gate positioned by carefully backgating blast cells for their primitive marker (CD34, CD117) expression. AML blasts may be positive or sometimes negative for CD34, CD117 expression. AML blasts are then scanned for aberrant LAIPs using DfN approach. LAIPs are identified using the principles of cross-lineage expression, asynchronous expression or over/under expression of markers.
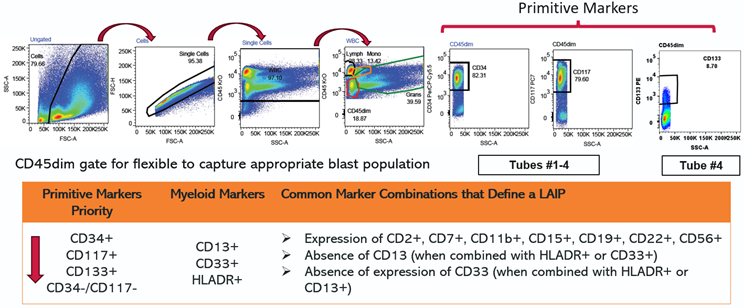
Figure 2. AML-MRD MFC Analysis (Gating) Paradigm
Method Performance Summary
The analytical validation focused on evaluation of sensitivity (lower limit of quantitation (LLOQ), MRD cut point), reproducibility, repeatability, and accuracy (orthogonal verification by morphological examination) as key criteria for assessing MFC method performance. The use of Bone Marrow Mononuclear Cell (BMMC) specimens was necessitated by difficulties with procuring adequate native samples with defined tumor burden, however, the BMMC experimental design is supported by clinical cytometry consortia (Wood et al., 2013) and represents a more challenging (worst-case) scenario due to potential antigen decay and cell loss. Specimen numbers were chosen based on recommendations by clinical cytometry consortia (Wood et al., 2013) and CLSI H62 for MFC methods (Wayne, 2020). The results of the AML-MRD MFC assay validation activities were found to be acceptable, with the MRD cutoff defined at 0.1%. Complete details are described in an independent validation report. We are currently supplementing the validation package with native specimen testing to meet evolving health authority requirements and obtain Investigational Use Only (IUO) label.
Discussion & Clinical Experience
Utilization of a core set of markers previously proven to correlate with survival outcomes indicate that the MRD results generated by Navigate BioPharma are suitable for interpretation of treatment efficacy. The sensitivity (MRD cut point) results indicate a lower limit of quantification of at least 0.1% LAIP/WBC across five independent patient LAIPs. The presence of non-leukemic hematopoietic precursors in healthy individuals with phenotypes overlapping LAIPs precluded reliable quantitation of disease burden below 0.1% except for one patient, whose LAIP could be reliably quantified at 0.01% due to absence of overlapping healthy donor phenotypes. Both the frequencies and the nature of LAIPs observed in healthy donors are within the range reported by others (Feller et al., 2004; Kern et al., 2003). As highlighted in Schuurhuis 2018, based on the large numbers of LAIPs that can be defined in AML (up to 100) and their inherent differences in specificity, cutoff levels that capture MRD positivity and are applicable to all LAIPs must be relatively high (i.e., 0.035% to 0.2%). A cutoff of 0.1% was found clinically relevant in published studies and is the recommended clinical cutoff by the ELN AML-MRD working party (Heuser et al., 2021). Thus, a 0.1% threshold to distinguish MRD-positive from MRD-negative patients was implemented with our method for clinical trials.
Table 3: Summary of Representative Patient Results
Primary Attributes | Myeloid & Lymphoid Cells | Tumor Phenotype & Content [LAIP or DfN] | |||
Parameter | Reported Result | Parameter | Reported Result | Parameter | Reported Result |
Disclaimer | NA | CD45dim Events | 122832 | LAIP-1_Phenotype | CD34+CD13+CD33- |
Tube 1 | Yes | CD45dim (Freq of WBC) | 29.33 | LAIP-1 Events | 13148 |
Tube 2 | Yes | CD34 Events | 97792 | LAIP-1 (Freq of CD34) | 11.74 |
Tube 3 | Yes | CD34 (Freq of CD45dim) | 79.61 | LAIP-1 (Freq of CD45dim) | 9.19 |
Tube 4 | Yes | CD34 (Freq of WBC) | 23.35 | LAIP-1 (Freq of WBC) | 2.38 |
Tube 5 | Yes | CD117 Events | 98489 | LAIP-2_Phenotype | CD34+CD13+HLADR- |
MRD Status | MRD Positive | CD117 (Freq of CD45dim) | 80.18 | LAIP-2 Events | 3499 |
Viability Description | Adequate | CD117 (Freq of WBC) | 23.52 | LAIP-2 (Freq of CD34) | 3.12 |
Primitive Marker | CD34+ | CD133 Events | 91092 | LAIP-2 (Freq of CD45dim) | 2.45 |
WBC Events | 418758 | CD133 (Freq of CD45dim) | 74.16 | LAIP-2 (Freq of WBC) | 0.63 |
Viability (Freq of WBC) | 90.39 | CD133 (Freq of WBC) | 21.75 | DfN-LAIP-1_Phenotype | CD34+CD13+CD22+ |
Hemodilution | No | Lymph (Freq of WBC) | 29.31 | DfN-LAIP-1 Events | 30425 |
|
| Mono (Freq of WBC) | 10.35 | DfN-LAIP-1 (Freq of CD34) | 27.04 |
Over the past decade, this validated method has been utilized for understanding the depth of tumor clearance and length of remission in multiple Phase 2 and Phase 3 clinical trials assessing the effect of immune checkpoint inhibitors in combination with venetoclax and azacitidine on MRD (see Table 4). MRD assessments are proposed to complement the efficacy results in these clinical studies (e.g., MRD negative CR rate), providing a measure of deeper responses with reported prognostic value of long-term outcome in AML (Freeman et al., 2018; Ivey et al., 2016; Jongen-Lavrencic et al., 2018; Terwijn et al., 2013). We believe incorporating MRD monitoring by MFC method in AML and High Risk MDS clinical trial programs can provide valuable insights into treatment response, patient risk stratification, and personalized therapeutic approaches, ultimately leading to improved patient outcomes.
Table 4: Summary of Clinical Trial Experience with AML-MRD MFC Assay
| Clinical Trial #1 | Clinical Trial #2 | Clinical Trial #3 | |
| Disease | MDS or CMML-2 | AML | AML |
| Study Population | Adult patients with intermediate or high risk MDS or CMML-2 | Adult patients unfit for chemotherapy | Patients with presence of MRD after allogenic stem cell transplantation |
| Phase | III | II | Ib/II |
| Investigational Agents | ICI Blocker w/ Azacitidine | ICI Blocker w/ Azacitidine + Venetoclax | ICI Blocker w/ Azacitidine |
| Specimen Type | PB and BM | PB and BM | BM |
| # of Cases Reported | 1540 | 438 | 135 |
References
Buccisano, F., Hourigan, C.S., and Walter, R.B. (2017). The Prognostic Significance of Measurable ("Minimal") Residual Disease in Acute Myeloid Leukemia. Curr Hematol Malig Rep 12, 547-556.
Cloos, J., Harris, J.R., Janssen, J., Kelder, A., Huang, F., Sijm, G., Vonk, M., Snel, A.N., Scheick, J.R., Scholten, W.J., et al. (2018). Comprehensive Protocol to Sample and Process Bone Marrow for Measuring Measurable Residual Disease and Leukemic Stem Cells in Acute Myeloid Leukemia. J Vis Exp.
Dohner, H., Weisdorf, D.J., and Bloomfield, C.D. (2015). Acute Myeloid Leukemia. N Engl J Med 373, 1136-1152.
Feller, N., van der Pol, M.A., van Stijn, A., Weijers, G.W., Westra, A.H., Evertse, B.W., Ossenkoppele, G.J., and Schuurhuis, G.J. (2004). MRD parameters using immunophenotypic detection methods are highly reliable in predicting survival in acute myeloid leukaemia. Leukemia 18, 1380-1390.
Freeman, S.D., Hills, R.K., Virgo, P., Khan, N., Couzens, S., Dillon, R., Gilkes, A., Upton, L., Nielsen, O.J., Cavenagh, J.D., et al. (2018). Measurable Residual Disease at Induction Redefines Partial Response in Acute Myeloid Leukemia and Stratifies Outcomes in Patients at Standard Risk Without NPM1 Mutations. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 36, 1486-1497.
Heuser, M., Freeman, S.D., Ossenkoppele, G.J., Buccisano, F., Hourigan, C.S., Ngai, L.L., Tettero, J.M., Bachas, C., Baer, C., Bene, M.C., et al. (2021). 2021 Update on MRD in acute myeloid leukemia: a consensus document from the European LeukemiaNet MRD Working Party. Blood 138, 2753-2767.
Hu, G.H., Cheng, Y.F., Lu, A.D., Wang, Y., Zuo, Y.X., Yan, C.H., Wu, J., Sun, Y.Q., Suo, P., Chen, Y.H., et al. (2020). Allogeneic hematopoietic stem cell transplantation can improve the prognosis of high-risk pediatric t(8;21) acute myeloid leukemia in first remission based on MRD-guided treatment. BMC cancer 20, 553.
Ivey, A., Hills, R.K., Simpson, M.A., Jovanovic, J.V., Gilkes, A., Grech, A., Patel, Y., Bhudia, N., Farah, H., Mason, J., et al. (2016). Assessment of Minimal Residual Disease in Standard-Risk AML. N Engl J Med 374, 422-433.
Jongen-Lavrencic, M., Grob, T., Hanekamp, D., Kavelaars, F.G., Al Hinai, A., Zeilemaker, A., Erpelinck-Verschueren, C.A.J., Gradowska, P.L., Meijer, R., Cloos, J., et al. (2018). Molecular Minimal Residual Disease in Acute Myeloid Leukemia. N Engl J Med 378, 1189-1199.
Keeney, M., Halley, J.G., Rhoads, D.D., Ansari, M.Q., Kussick, S.J., Karlon, W.J., Mehta, K.U., Dorfman, D.M., and Linden, M.A. (2015). Marked Variability in Reported Minimal Residual Disease Lower Level of Detection of 4 Hematolymphoid Neoplasms: A Survey of Participants in the College of American Pathologists Flow Cytometry Proficiency Testing Program. Archives of pathology & laboratory medicine 139, 1276-1280.
Kern, W., Danhauser-Riedl, S., Ratei, R., Schnittger, S., Schoch, C., Kolb, H.J., Ludwig, W.D., Hiddemann, W., and Haferlach, T. (2003). Detection of minimal residual disease in unselected patients with acute myeloid leukemia using multiparameter flow cytometry for definition of leukemia-associated immunophenotypes and determination of their frequencies in normal bone marrow. Haematologica 88, 646-653.
Lowenberg, B., Pabst, T., Maertens, J., Gradowska, P., Biemond, B.J., Spertini, O., Vellenga, E., Griskevicius, L., Tick, L.W., Jongen-Lavrencic, M., et al. (2021). Addition of lenalidomide to intensive treatment in younger and middle-aged adults with newly diagnosed AML: the HOVON-SAKK-132 trial. Blood Adv 5, 1110-1121.
Ossenkoppele, G.J., and Schuurhuis, G.J. (2014). MRD in AML: it is time to change the definition of remission. Best Pract Res Clin Haematol 27, 265-271.
Schuurhuis, G.J., Heuser, M., Freeman, S., Bene, M.C., Buccisano, F., Cloos, J., Grimwade, D., Haferlach, T., Hills, R.K., Hourigan, C.S., et al. (2018). Minimal/measurable residual disease in AML: a consensus document from the European LeukemiaNet MRD Working Party. Blood 131, 1275-1291.
Short, N.J., Rytting, M.E., and Cortes, J.E. (2018). Acute myeloid leukaemia. Lancet 392, 593-606.
Short, N.J., Zhou, S., Fu, C., Berry, D.A., Walter, R.B., Freeman, S.D., Hourigan, C.S., Huang, X., Nogueras Gonzalez, G., Hwang, H., et al. (2020). Association of Measurable Residual Disease With Survival Outcomes in Patients With Acute Myeloid Leukemia: A Systematic Review and Meta-analysis. JAMA oncology 6, 1890-1899.
Terwijn, M., van Putten, W.L., Kelder, A., van der Velden, V.H., Brooimans, R.A., Pabst, T., Maertens, J., Boeckx, N., de Greef, G.E., Valk, P.J., et al. (2013). High prognostic impact of flow cytometric minimal residual disease detection in acute myeloid leukemia: data from the HOVON/SAKK AML 42A study. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 31, 3889-3897.
Tettero, J.M., Dakappagari, N., Heidinga, M.E., Oussoren-Brockhoff, Y., Hanekamp, D., Pahuja, A., Burns, K., Kaur, P., Alfonso, Z., van der Velden, V.H.J., et al. (2023). Analytical assay validation for acute myeloid leukemia measurable residual disease assessment by multiparametric flow cytometry. Cytometry B Clin Cytom 104, 426-439.
Wayne (2020). Validation of Assays Performed by Flow Cytometry (1st ed. CLSI document H62).
Wood, B., Jevremovic, D., Bene, M.C., Yan, M., Jacobs, P., Litwin, V., and Group, I.I.W. (2013). Validation of cell-based fluorescence assays: practice guidelines from the ICSH and ICCS - part V - assay performance criteria. Cytometry B Clin Cytom 84, 315-323.